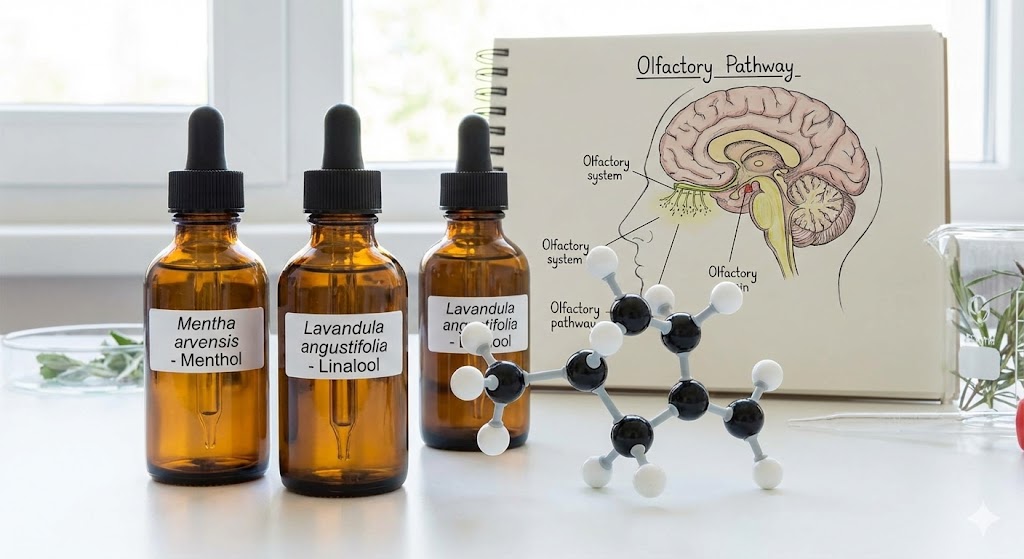
When you inhale the sharp scent of peppermint or the calming aroma of lavender, you aren't just smelling a pleasant fragrance—you are experiencing a complex chemical reaction. Essential oils are often called the "life force" of plants, but scientifically, they are volatile organic compounds (VOCs). These are potent chemical powerhouses that plants produce to protect themselves from pests and bacteria.
What makes essential oils so effective for humans is their molecular size. The compounds in these oils—like menthol in mint or linalool in lavender—are lipid-soluble and incredibly small. This allows them to pass easily through biological barriers, including our skin and cell membranes.
How do they work? There are two main pathways:
Inhalation: When you breathe in an essential oil, the molecules interact with the olfactory bulb in your nose, which has a direct hotline to the limbic system in the brain—the area responsible for emotions and memory. This is why a whiff of mint can instantly make you feel more alert.
Absorption: When applied topically (diluted), these small molecules penetrate the skin layers and enter the bloodstream, delivering their therapeutic properties system-wide.
In an era of synthetic fragrances, the science of genuine essential oils stands apart. It relies on the purity of the extraction process—usually steam distillation—to ensure that the delicate chemical profile of the plant remains intact. Whether used in pharmaceuticals or aromatherapy, essential oils represent the perfect marriage of ancient botany and modern chemistry.